2022 RSC Medicinal Chemistry Emerging Investigator Lectureship
Nominations are now open for the 2022 RSC Medicinal Chemistry Emerging Investigator Lectureship and close on 08 October 2021. The lectureship is open to candidates who received their PhD in 2012 or later and who have made a significant contribution to medicinal chemistry in their early career.
The RSC welcome nominations from anyone, to be sent to our journal inbox at medchem-rsc@rsc.org, but ask that nominations include the name and affiliation of the researcher, along with at least one paragraph explaining their achievements and why they should be considered (a CV is not required, but is helpful if sent along with the nomination). Additionally, self-nominations are welcomed if accompanied by a letter of support from the nominees’ institute.
There are more details about nominations on our webpage here: https://www.rsc.org/journals-books-databases/about-journals/rsc-medicinal-chemistry/#Lectureship_MChemCom.
21st RSC / SCI Medicinal Chemistry Symposium
Registration for the 21st RSC / SCI Medicinal Chemistry Symposium Monday-Wednesday, 13th-15th September 2021 hosted at Churchill College, Cambridge, UK is now open. Twitter hashtag - #CamMedChem21
As usual there is a stella lineup of presentations and there is still time for some submissions.
I'd like to highlight one talk that is close to my heart. Ed Griffen will talking about The COVID Moonshot: SARS-CoV2 oral antiviral therapeutics from an Open Science global collaboration.
The COVID Moonshot is an ambitious crowdsourced initiative to accelerate the development of a COVID antiviral. We work in the open with no intellectual property constraints. This way, any scientist can view submitted drug designs and experimental data to inspire new design ideas. We use our cutting-edge machine learning tools and Folding@home's crowdsourced supercomputer to determine which drug designs to send to our partners to make and test in the lab. With each drug design tested, we get closer to our goal.
This talk will be in the late breaker session and because of the open nature of the project it will be a chance to really see the very latest results "hot off the press".
You can have a look at the latest results here now. https://covid.postera.ai/covid.
30th April: Late breaker and early poster deadline
23rd July: Final poster deadline
Registration link https://www.maggichurchouseevents.co.uk/bmcs/cmc21/index.htm
RSC BMCS Hall of Fame
The RSC Biological and Medicinal Chemistry initiated a Hall of Fame a short while ago and I'd like to highlight the relevant page of the BMCS website.
The Hall of Fame is to recognise prominent chemists for outstanding, sustained, contributions to any area of interest to the BMCS, eg medicinal chemistry, agriscience, biooorganic chemistry, chemical biology. This is an Individual award to recognise prominence and significant, sustained, scientific impact in the field of medicinal chemistry, agriscience or chemical biology, including teaching excellence, outstanding contributions to the BMCS, or any combination thereof.
The first inductee to the BMCS Hall of Fame was Professor C Robin Ganellin FRS, Emeritus Professor of Medicinal Chemistry at University College London. He co-discovered histamine H2-receptors with James Black and co-invented the anti-ulcer drug cimetidine. He co-discovered butabindide, an inhibitor of the enzyme tripeptidyl peptidase II, and co-invented the histamine H3-receptor antagonist drug, pitolisant.
Cimetidine was the first histamine H2 receptor antagonist drug that inhibits stomach acid production and used in the treatment of heartburn and peptic ulcers.
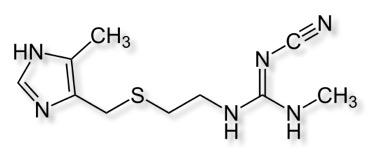
Oral bioavailability is 65% and it has a half-life of 2 hours.
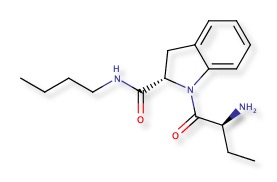
Butabindide is an inhibitor of the enzyme tripeptidyl peptidase II designed as an anti-obesity drug.
The histamine H3-receptor antagonist Pitolisant, is used for the treatment of excessive daytime sleepiness (EDS) in adults with narcolepsy
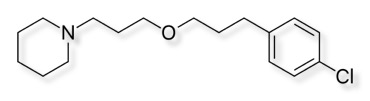
Pitolisant is well absorbed (90%) and has an elimination half-life of 10-12 hours.
The 2019 inductee was Sir Simon Campbell CBE FRS FMedSci who is probably best known for his work leading to Doxazosin, for high blood pressure and angina and Amlodipine – used to treat high blood pressure and prostrate enlargement.
Doxazosin is a α1-selective adrenergic blocker in the quinazoline class of compounds
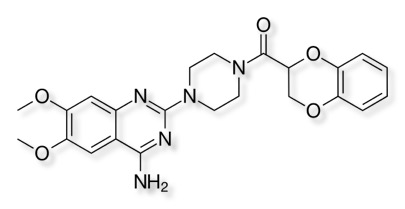
Oral bioavailability is 65% and elimination half-life 22 hours, , it highly plasma protein bound (98%). Hepatic metabolism of doxazosin produces inactive O-demethylated and C-hydroxylated metabolites.
Amlodipine is a long acting calcium channel antagonist, it blocks L-type calcium channels in muscle cells and N-type calcium channels in the central nervous system.
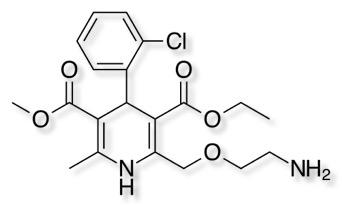
Amlodipine is well absorbed by the oral route with a mean oral bioavailability around 60%; the half-life of amlodipine is about 30 h to 50 h, it highly plasma protein bound (97.5%). Renal elimination is the major route of excretion with about 60% of an administered dose recovered in urine, largely as inactive pyridine metabolites. Amlodipine is on the World Health Organisation's List of Essential Medicines.
31st symposium on Medicinal Chemistry in Eastern England
The Symposium on Medicinal Chemistry in Eastern England, known colloquially as the "Hatfield MedChem" meeting, is a highly successful, long-standing, one-day meeting which runs annually. The scientific program comprises of presentations showcasing medicinal chemistry case studies from tools to candidates, across a range of modalities, therapeutic areas and target classes, as well as covering more general topics from the forefront of drug discovery of relevance to medicinal chemists. The meeting aims to be informal and interactive and the event will offer excellent scientific and networking opportunities for all those working in medicinal chemistry and drug discovery.
It will take place on Thursday 30th April 2020 at The Fielder Centre, Hatfield, Hertfordshire, UK
Registration is now open.
Full details of the scientific programme and registration details are on the website https://www.maggichurchouseevents.co.uk/bmcs/hatfield_symposium-31.htm
Always a very popular meeting so registration early is recommended.
Twitter hashtag #HatfieldMedChem20
EFMC Prize for a Young Medicinal Chemist in Industry/Academia
I just thought I'd highlight this award.
The EFMC created the “EFMC Prize for a Young Medicinal Chemist in Industry/Academia” as we felt it was important to acknowledge and recognise outstanding young medicinal chemists (≤ 12 years after PhD) working in European industry and academia. The 2020 Prizes will be given at the XXVI EFMC "International Symposium on Medicinal Chemistry" (EFMC-ISMC 2020) to be held in Basel, Switzerland on September 6-10, 2020. Both prizes consist of a diploma, an invitation to give an oral communication at the EFMC-ISMC, and a cash prize of € 1,000.
To find out more on the regulations and the application procedure visit the EFMC website: https://www.efmc.info/prizes, closing date Jan 31 2020.
Early career MedChem workshop
The Early Career MedChem Workshop is a pre-meeting workshop taking place on the Sunday afternoon before the 20th SCI/RSC Medicinal Chemistry Symposium, Churchill College, Cambridge, Sunday 8 - Wednesday 11 September 2019.
It is aimed at early career (up to 5 years’ experience) medicinal chemists. Registration for this event will be at no additional cost to the main meeting.
The workshop will consist of a team-based exercise around a virtual medicinal chemistry programme and will offer participants:
- Training in advanced medicinal chemistry
- Testing out leadership/decision making skills
- Networking between potential future leaders of different organisations
- Introductions of possible future collaborators from pharma, CRO, charity and SME sectors
- Opportunity for informal interaction with experienced medicinal chemists workshop facilitators who will be open to questions around their own experiences
- Attendance at the full 20th SCI/RSC Medicinal Chemistry Symposium including lectures on a full range of drug targets, key enabling processes and technologies
Feedback from previous events has been excellent for this unique learning experience.
Also note there are student bursaries to help fund attendance at this event, email conferences@soci.org for more information.
Upcoming Conferences
I just thought I'd mention a couple of meetings I'm helping to organise.
2nd RSC-BMCS / RSC-CICAG Artificial Intelligence in Chemistry
Artificial Intelligence is presently experiencing a renaissance in development of new methods and practical applications to ongoing challenges in Chemistry. Following the success of the inaugural “Artificial Intelligence in Chemistry” meeting in 2018 a second meeting has been organised at Fitzwilliam College, Cambridge (2nd to 3rd September 2019). The lineup is now finalised and looks like a great selection of speakers. There is still time to submit posters (closing date 5th July).
Registration is open and there are discounts for RSC members.
The Twitter hashtag - #AIChem19 is already being actively used.
20th SCI/RSC Medicinal Chemistry Symposium
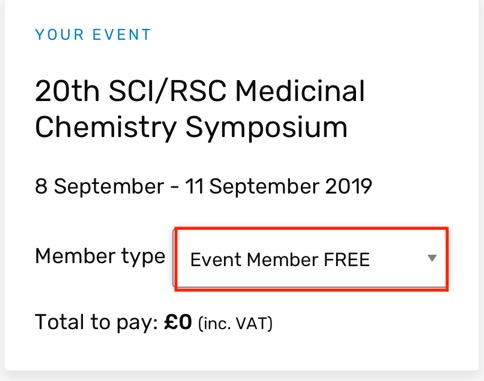
This is Europe’s premier biennial Medicinal Chemistry event, focussing on first disclosures and new strategies in Medicinal Chemistry. It takes place a Churchill College, Cambridge UK, 8 September - 11 September 2019. There is a fantastic lineup of speakers and looks to be one of the highlights of the MedChem calendar. Early career scientists can also take part in a Medicinal chemistry workshop on the Sunday afternoon, a great way for people to learn medicinal chemistry and meet other scientists in a fun and informal setting.
You can register here both RSC and SCI members get a reduced rate, and despite the slightly confusing page on the SCI website you don't have to be a member to attend, just select "Event Member FREE from the dropdown menu and you can register for the event without membership.
Twenty Years of the Rule of Five
It has been over twenty years since Lipinski published his work determining the properties of drug molecules associated with good solubility and permeability. Since then, there have been a number of additions and expansions to these “rules”. There has also been keen interest in the application of these guidelines in the drug discovery process and how these apply to new emerging chemical structures such as macrocycles.
This meeting aims to have a look at the impact the Ro5 has had on drug discovery and as well as looking to the future and how we use these rules in the changing drug compound landscape as drug discovery moves into novel areas of chemistry.
There is a very exciting group of speakers and the timetable has been designed to allow a panel discussion after each session. Given the topic and the speakers I'm sure these will be entertaining sessions.
You can register here and there are discounts for RSC members
Twitter hashtag - #RuleofFive2019
Early Career MedChem Workshop
One of the highlights of the SCI/RSC Cambridge MedChem Meetings the Early Career MedChem Workshop, a satellite workshop consisting of a team-based exercise around a virtual medicinal chemistry programme will be held on the Sunday afternoon prior to this event, aimed at early career (up to 5 years’ experience) medicinal chemists. Registration for this event will be at no additional cost to the main meeting, Read the flyer here.

Registration is on the 20th SCI / RSC Medicinal Chemistry Symposium website
20th Cambridge MedChem meeting Registration open
I know the is eagerly anticipated because I have folder of email enquiries I can now respond to.
Registration for the 20th Cambridge MedChem Meeting is now open !!
https://www.soci.org/events/20th-scirsc-medicinal-chemistry-symposium.
Really is a fabulous lineup of speakers. Also includes Malcolm Campbell Memorial Prize presentation
20th Cambridge MedChem meeting
On 8-11 September 2019 we will be holding the 20th Cambridge Medicinal Chemistry Symposium, this marks a significant milestone for this meeting and the committee would like to use the opportunity to highlight some of the first disclosures of now important medicines and other events from the 40 year history of this meeting.
We have all the programmes from the 13th to 19th meetings, and I've managed to get details of the first meeting shown in this PDF. If you could share scanned copies of programme from the 2nd to 12th meetings it would be really great.
Also if you have particular memories of meetings you would be willing to share feel free to send an email.
RSC Medicinal Chemistry Residential School 2019
The Royal Society of Chemistry Medicinal Chemistry Residential School takes place 2 - 7 June 2019, Loughborough, United Kingdom is a fantastic opportunity for anyone starting out or contemplating a career in Drug Discovery.
The school is designed for graduate and post-doctoral chemists with 1-5 years’ experience in the field of drug research. Drug discovery is an interdisciplinary subject so delegates from biological or computational backgrounds will benefit from attendance at the school. In addition, final year PhD students from pharmaceutical or organic chemistry contemplating a career in drug discovery are also encouraged to attend.
The course includes the following topics:
- Target Validation
- Computational Chemistry
- Biological Mechanisms
- Pharmacokinetics and Drug Metabolism
- Screening of New Compounds
- Patents
- Molecular Biology in Medicinal Chemistry
- Exploiting a Chemical Lead
- Combinatorial Chemistry and Molecular Diversity
- Case Histories of Drug Discovery
- Toxicology in Drug Discovery
- Pharmaceutical Considerations in Drug Development
- Structure-guided Drug Design
- Physical Properties and Quantitative Structure-Activity Relationships
- Hints and Tips in Medicinal Chemistry
Full details here http://www.rsc.org/events/detail/33379/medicinal-chemistry-residential-school-2019.
2019 Medicinal Chemistry Residential School
Registration for the RSC 2019 Medicinal Chemistry Residential School is now open, it takes place in Loughborough, UK 2-7 June 2019.
Through an in-depth programme of lectures, case studies and hands-on tutorial sessions, this five-day course strengthens excellence in medicinal chemistry by advancing understanding of the factors governing modern drug discovery. Full details are here.
Make sure that you register for this course as soon as possible to take full advantage of early bird savings. Registration includes attendance at all sessions, refreshments and lunch on each full day – plus a conference dinner with wine on Thursday 6 June.
20th SCI/RSC Medicinal Chemistry Symposium
The first circular for the 20th SCI/RSC Medicinal Chemistry Symposium (aka the Cambridge MedChem Meeting) has been announced. This is also a call for abstracts for both oral and poster submissions. Potential contributions should be communicated to the secretariat at conferences@soci.org by Friday 9th November 2018.

You can download the full details of the meeting here PDF.
A number of outstanding talks have already been confirmed.

29th Symposium on Medicinal Chemistry in Eastern England
The Symposium on Medicinal Chemistry in Eastern England, known colloquially as the "Hatfield MedChem" meeting, is a highly successful, long-standing, one-day meeting which runs annually will be held 26 April 2018 09:30-17:00, Hatfield, United Kingdom
The Organising Committee are inviting abstract submissions for both oral and poster presentation. Please download an abstract template from the event website and return it the secretariat. Closing dates for submissions are:
24th November 2017 for oral abstracts
23rd February 2018 for poster abstract
The BMCS Mastering MedChem III: 3rd RSC-BMCS symposium on mastering medicinal chemistry
This meeting is aimed at all those who wish to become better drug hunters and heed warnings from the past. (22 March 2017 09:00-19:00, Cardiff Bay, United Kingdom).
In the main there are two types of drug discovery programmes: those that hit serious problems and those that are going to hit serious problems. The difference between success and failure is how we, as medicinal chemists, tackle and resolve the problems
Sounds a great meeting both for those starting out in their careers and for those looking to pick up new tips.
28th symposium on Medicinal Chemistry in Eastern England
28th symposium on Medicinal Chemistry in Eastern England, Thursday 27th April 2017, The Fielder Centre, Hatfield, Hertfordshire, UK
Organised by RSC-BMCS (Royal Society of Chemistry – Biological and Medicinal Chemistry Sector)
Programme
09.00 Registration, refreshments and exhibition
Session chair: Adrian Hall, UCB
09.30 Opening remarks
Nicole Hamblin, GlaxoSmithKline
09.35 From phenotypic hit to a validated target for tuberculosis
Robert Bates, GlaxoSmithKline
10.10 Discovery of potent inhibitors of the lysophospholipase autotaxin
Prit Shah, Cancer Research Technology
10.45 Refreshments and exhibition
11.15 Development of Tesirine: a clinical antibody-drug conjugate pyrrolobenzodiazepine payload: medicinal chemistry at the frontier between small molecules and biologics
Arnaud Tiberghien, Spirogen
11.50 NMR conformational analysis in molecular design – case studies and impact
Martin Packer, AstraZeneca
12.25 Highly potent cell-penetrant inhibitors of the KEAP1-NRF2 protein-protein interaction via X-ray fragment screening
Charlotte Griffiths-Jones, Astex Pharmaceuticals
13.00 Lunch and exhibition
Session Chair: Simon Ward, University of Sussex
14:05 Selective on-target chemical probes of protein-protein interactions
Alessio Ciulli, University of Dundee
14.40 Solid state studies of a preclinical candidate in a CRO environment: the importance of de-risking early
Russell Scammell, Charles River
15.15 Refreshments and exhibition
15.45 Drug discovery case study
Tom Miller, Shire
16.20 Optimisation of a series of novel smoothened inhibitors
Matilda Bingham, RedX
16.55 Concluding remarks
17.00 Close
19th RSC/SCI Medicinal Chemistry Symposium
I’m delighted to highlight the first announcement of the 19th RSC/SCI Medicinal Chemistry Symposium to be held in Cambridge in September 2017. Europe’s premier biennial Medicinal Chemistry event, focusing on first disclosures and new strategies in medicinal chemistry.
Open Source Malaria online meeting
The next strategy meeting for the Open Source Malaria team is on May 24th. This is great chance to find out more about this effort and to look for opportunities where you might be able to contribute
Outline agenda:
- Project background & scientific objectives (5)
- Overview of project information sources - ie what's where (5)
- Data sources & summary of analyses (5-10)
- SAR overview, questions for the audience (20)
- Discussion (10-15)
- Final guidance on what's required from the audience & how to submit your suggestions & what happens next (5)

You can register here https://attendee.gotowebinar.com/register/7722880987090170883 - you need to register in order to receive the link for the meeting itself. Please forward this on to anyone you think might be interested,
The Handbook of Medicinal Chemistry
The Handbook of Medicinal Chemistry is a new book providing insight and advice for medicinal chemists.
Drug discovery is a constantly developing and expanding area of research. Developed to provide a comprehensive guide, the Handbook of Medicinal Chemistry covers the past, present and future of the entire drug development process. Highlighting the recent successes and failures in drug discovery, the book helps readers to understand the factors governing modern drug discovery from the initial concept through to a marketed medicine. With chapters covering a wide range of topics from drug discovery processes and optimization, development of synthetic routes, pharmaceutical properties and computational biology, the handbook aims to enable medicinal chemists to apply their academic understanding to every aspect of drug discovery. Each chapter includes expert advice to not only provide a rigorous understanding of the principles being discussed, but to provide useful hints and tips gained from within the pharmaceutical industry. This expertise, combined with project case studies, highlighting and discussing all areas of successful projects, make this an essential handbook for all those involved in pharmaceutical development.
A free app has also been created in collaboration with the editors of the book. The Medicinal Chemistry Toolkit provides a suite of resources to support the day to day work of a medicinal chemist
Free online MedChem course
I’ve just been sent details of a new medicinal chemistry course.
Medicinal Chemistry: The Molecular Basis of Drug Discovery
This course explores how to bring a drug from concept to market, and how a drug's chemical structure relates to its biological function. The course opens with an introduction to the drug approval process. This introduction combines the social, economic, and ethical aspects of drug discovery. Topics include how diseases are selected for treatment, the role of animal testing, and the costs of various discovery phases. The course then focuses on the scientific side of drug discovery. Topics include how drugs interact with biological molecules, drug absorption and elimination, and the discovery of weakly active molecules and their optimization into viable drugs.
The course starts 10 March, it is estimated the course will require 6-8 hours per week and runs for 7 weeks. The course was organised by Erland Stevens who wrote the medchem textbook Medicinal Chemistry: The Modern Drug Discovery Process.
Biological and MedChem Meeting
I just got details of an interesting meeting in Cambridge, UK later this year.


