The European Lead Factory WORKS!
The European Lead Factory (ELF) is a collaborative public-private partnership aiming to deliver novel lead molecules for drug discovery programs. When the consortium was formed around 5 years ago there was a lot of scepticism about whether a group of 30 partners rating from large Pharma companies to small academic groups could ever agree on a legal framework that would allow the ELF to function. In a addition, in an industry where confidentiality was critical to maintaining intellectual property the idea that a group of large Pharma companies would share their sample collections often regarded as the "Crown Jewels" seemed impossible. However I was at the European Lead factory Stakeholder Meeting (24-25 April 2017) and it is clear that is has been a success.
The original compound collection was made up of 300,000 compounds provided by the major pharma partners (EFPIA) from their in house collections, this has been supplemented by nearly 200,000 molecules have been added by a bespoke synthetic chemistry programme; which was carefully selected for novelty, drug-like properties, diversity and synthetic tractability. These different collections now form the Joint European Compound Library (JECL). Now that the screening centre has been running for a number of years we can look back and get a more accurate view of the quality of the molecules in the library. The results show that the fears the EFPIA partners might simply dump uninteresting compounds into the collection are completely unfounded. Hit rates for the for the ELF run screens are similar to those that companies have run in house, in addition QC analysis of the hits from the screens confirms high purity and confirmed structures. Subsequent resynthesis of hits recapitulated the original screening data, again underlining the quality of the compound collection. Whilst there is less screening information about the newly synthesised compounds the evidence to date suggests these new compounds are enhancing the collection whilst maintaining the high levels of quality.

The uHTS robot used for screening academic and SME target programmes. Courtesy of Pivot Park Screening Centre.
The combination of collections from multiple sources has also given a unique opportunity to compare collections. It turns out there is minimal overlap between the different collections, which just serves to underline that chemical space is very, very large. The new compounds have been designed to further explore chemical space and there is certainly the opportunity for continuing to build on this to further expand the collection. Overall the hit rates from the different collections are identical as is compound quality. Perhaps more importantly several partners reported hits for projects that had failed provide leads when screened against their own internal libraries.
As the screening resources have been developed they have brought in orthogonal assays to provide evidence of target engagement such as SPR or Dynamic mass redistribution DOI. This offers an extra level of validation for the hit compounds.
Access to the ELF has raised a few issues, whilst the Structural Genomics Consortium (SGC) group at the University of Oxford provides protein production and crystallography to the ELF many academics don't have access to the facilities or the know-how to develop a uHTS assay, fortunately funding agencies have recognised the unique importance of the opportunity the ELF presents and have provided the relatively modest funding required for the development of high quality, robust assays with a good enough window of signal relative to the noise to be suitable for identifying potentially a handful of active compounds among 100,000s of inactive compounds.
In addition to providing screening there is also the opportunity to continue the collaboration with the ELF to further develop these initial hits with additional studies funded by IMI (like resynthesis, DMPK, crystallography) which can lead to an ‘Improved Hit List’ (IHL), this is particularly valuable to academic groups who might not have access to medicinal chemistry expertise.
So who is using the ELF
So far nearly 150 proposals for screening have been received and as the map below shows, proposals have been received from a very wide geographic area from both academia and SMEs.
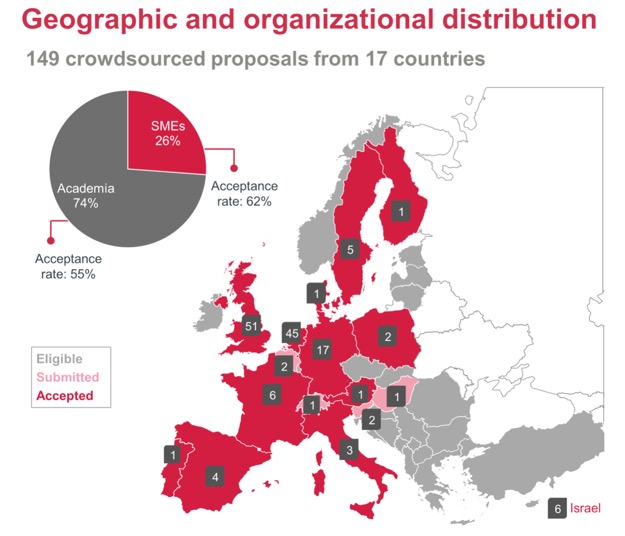
Image courtesy of P Kramer-Verhulst, T Sliedrecht, J S B de Vlieger, K M Orrling, and T Rijinders, ELF Programme Office, Lygature, The Netherlands.
For information on the eligible countries see http://ec.europa.eu/research/participants/data/ref/h2020/grantsmanual/hi/3cpart/h2020-hi-list-acen.pdf
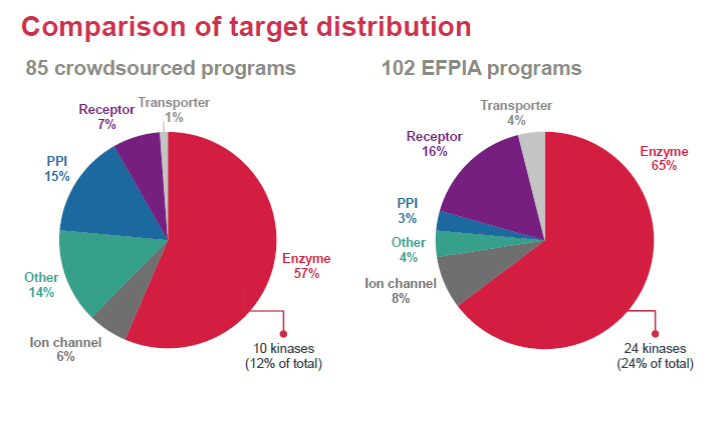
A wide variety of molecular targets have been covered, including enzymes, GPCR, Transporters, Ion Channels and protein-protein interactions, with enzymes being the most popular target.
Has the ELF been a success?
Measuring "success" in drug discovery is often troublesome, the bottom line is always marketed drugs, unfortunately that is a metric that can take a decade to come to fruition and since the first qualified hit list (QHL) was delivered in May 2014 we will have to wait a while yet. I think the ELF can offer a few alternative measures that might be important.
It was clear in the early discussions that this sort of open innovation was met with some trepidation, however now this has been replaced by a level of trust between the multiple partners. All would now be much more open to participating in this sort of collaboration in the future, which has to be good for the industry.
Access to a high quality sample collection together with infrastructure needed to run uHTS has been critical in moving a number of academic/small company projects forward and for providing leads for major Pharma when internal resources were unable.
Parkinson’s UK and the University of Sheffield have launched a joint venture biotech company, Keapstone Therapeutics. Parkinson’s UK has allocated 1 million GBP over the next sixteen months to further develop compounds that boost the internal cellular defence mechanisms against oxidative stress. These compounds were discovered by European Lead Factory.
Professor Chris Schofield at the University of Oxford, is collaborating with the European Gram-Negative Antibacterial Engine (ENABLE) project to further progress this programme towards clinical development. The Oxford team had the ELF screen their potential target against the JECL, the high throughput screen and initial triaging provided 50 qualified hits.
Dr. Margit Mahlapuu at the University of Gothenburg, one of the academic researchers who has benefited from EU Lead Factory screening activities by identifying a drug candidate series for a new target which could be used to reverse metabolic complications in type 2 diabetes. Dr. Mahlapuu went on to create a spin-out company based on these results together with Swedish state-owned GU Venture.
These success stories come from the very first public screens run by the ELF and emphasise the importance of the collections contributed by the EFPIA providing an industry standard quality screening deck.
In addition to the participation of the companies and academics a number of charities and Venture capitalists have become engaged, one of the successes of the ELF must surely be that VC are now willing to pick up very early stage projects.
What's Next?
There are still interesting chemical libraries that have not been explored, these could be used to expand the chemical space covered further. I know some charitable bodies have their own internal screening collections, perhaps these could be combined with the JECL in return for access to the combined collection?
The ELF is now a critical component in providing high quality hits for novel targets, however this has exposed other bottlenecks. The development of robust screening assays could be improved and a dedicated funding process would help. Whilst the medicinal chemistry is available to optimise the screening hits and turn them into well validated leads there is a critical need to provide complementary in vivo proof of concept (POC) studies. For many potential funders a novel target with a collection of lead molecules is of only modest interest without the POC and a PK/PD relationship consistent with the proposed mechanism.
The ELF have now released a video of the meeting.
Update (2 Jan 2018).
It has just been announced that the Joint European Compound Library (JECL) has now reached the target of 500,000 compounds.
So far, >180,000 novel compounds have been synthesised and the five library production partners Edelris, Mercachem, Sygnature, Syncom and Taros are well under way to deliver another ~20,000 compounds before the end of the project.
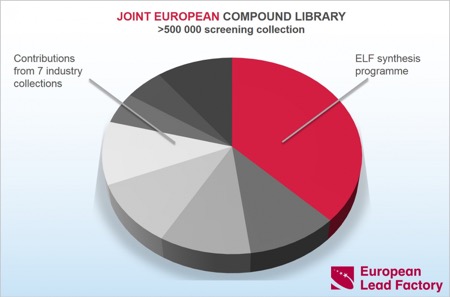
Only compounds meeting JECL quality criteria for purity (LC–MS purity >85%, average 97%) and quantity (>5 μmol, average 15 µmol) are added to JECL.
Last updated 3 January 2018